HTLV-1 Latency and Persistence Versus Host Immunity
Research summary
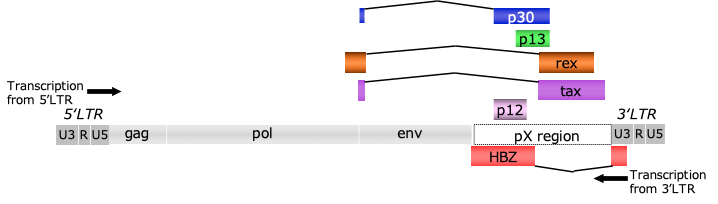
HTLV-1 (Human T-cell Leukemia Virus Type 1) is a retrovirus that primarily infects CD4-positive T cells, which are responsible for immunity. It is transmitted through breast milk, sexual contact, and blood transfusions. Once infected, it remains in the body for a long period. Most infected individuals remain asymptomatic, but some develop adult T-cell leukemia (ATL) or HTLV-1-associated myelopathy/tropical spastic paraparesis (HAM/TSP), a chronic inflammatory condition of the spinal cord. HTLV-1 is characterized by its ability to evade the immune system while replicating within infected cells, potentially causing disease over many years. In Japan, infection rates are known to be particularly high in the Kyushu and Okinawa regions.HTLV-1 possesses a single-stranded RNA genome approximately 10 kb in length. After infection, it is converted into DNA by reverse transcriptase and integrated into the host cell’s genome. Long terminal repeats (LTRs), which control transcription, are located at both ends of the genome. The central region contains the core structural genes: gag (structural proteins), pol (reverse transcriptase, etc.), and env (envelope proteins). A further distinctive feature is the pX region, a cluster of regulatory genes encoding proteins such as Tax and HBZ. These play crucial roles in viral replication, controlling the proliferation of infected cells, and evading the immune system. This complex genetic architecture underpins HTLV-1’s persistent infection and disease pathogenesis.
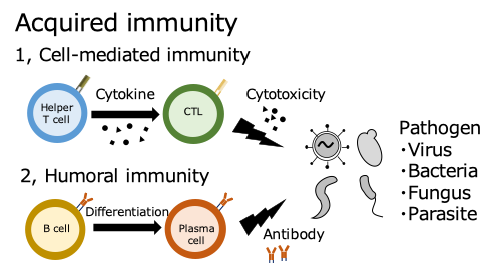
When a pathogen enters the body, immune cells first detect and engulf it, then communicate its characteristics (antigens) to other cells. In lymph nodes, T cells receive this information, become activated, multiply, and transform into functional cells. Helper T cells assist B cells, which then produce antibodies to attack the pathogen. Cytotoxic T cells directly destroy infected cells. Even after the infection subsides, some T- and B cells remain in the body as memory cells. This allows for a faster and stronger response if the same pathogen re-enters the body.
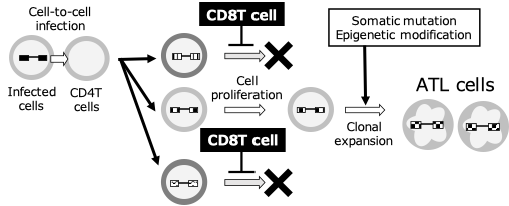
HTLV-1-infected CD4 T cells are recognized by the immune system as “abnormal cells” within the body. Within the infected cells, viral proteins are produced, and fragments of these are presented on the cell surface. CD8-positive T cells (killer T cells) are responsible for detecting these. T cells activate when their T cell receptors (TCR) recognize specific virus-derived peptide fragments on human leukocyte antigen (HLA) molecules. They then directly attack and eliminate infected cells. They also secrete substances called cytokines to assist other immune cells and enhance the antiviral response. In ATL as well, CD8 T cells targeting HTLV-1-derived antigens (e.g., Tax) exist and are thought to be crucial for controlling infected cells before they become tumorous. However, prolonged antigen stimulation causes CD8 T cells to become functionally depleted (exhausted), reducing their proliferative and cytotoxic capabilities. Furthermore, tumor cells evade the immune system by reducing antigen expression and increasing the expression of immunosuppressive molecules. Consequently, the surveillance mechanism mediated by CD8 T cells breaks down, leading to the onset and progression of ATL.
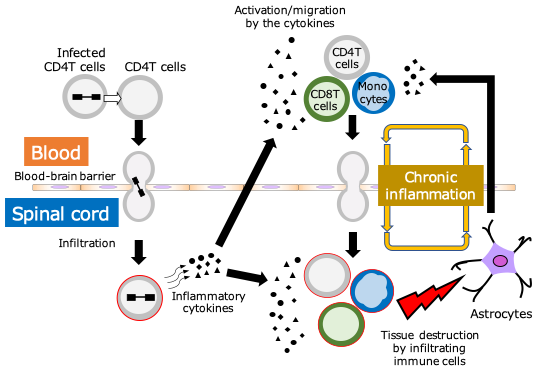
In HTLV-1-associated myelopathy/tropical spastic paraparesis (HAM/TSP), CD8 T cells recognizing HTLV-1 antigens (particularly Tax) increase in peripheral blood and cerebrospinal fluid, exhibiting strong cytotoxic activity and inflammatory cytokine production. These CD8 T cells attempt to eliminate infected T cells, but excessive and persistent activation causes them to infiltrate the spinal cord and cause inflammation in the surrounding nerve tissue. In particular, the production of IFN-γ and TNF-α activates glial cells, leading to chronic tissue damage. This results in symptoms such as spastic paralysis of the lower limbs and urinary dysfunction. In other words, CD8 T cells are ambiguous entities that are deeply involved in both virus control and disease formation.