Discovery of an intragenic viral silencer that enforces HTLV-1 latency via RUNX complex recruitment
A cis-regulatory “silencer” element within the HTLV-1 proviral genome suppresses viral transcription and supports long-term persistence.
Key points
- We identified a previously unrecognized intragenic cis-regulatory region in the HTLV-1 provirus that functions as a transcriptional silencer.
- Disruption of this silencer increases viral activity, suggesting a direct role in maintaining latency and influencing immune recognition.
- The RUNX transcription factor complex is recruited to this silencer and suppresses HTLV-1 transcriptional bursts.
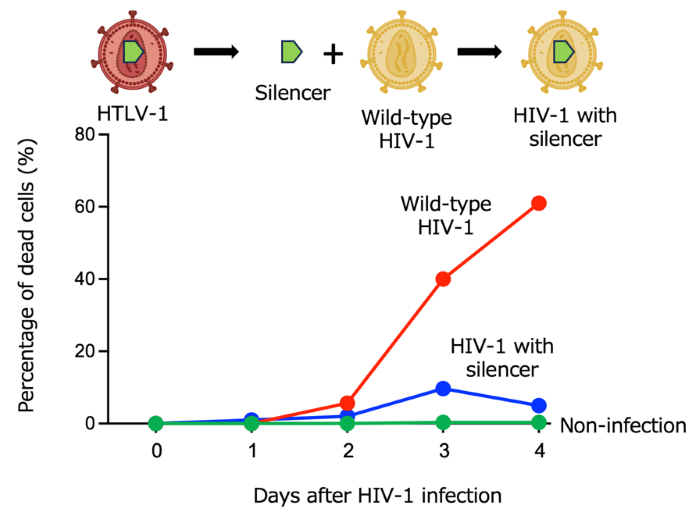
- When the HTLV-1 silencer module is introduced into HIV-1, it reduces virus production, indicating transferability of latency-enforcing circuitry across retroviruses.
Background
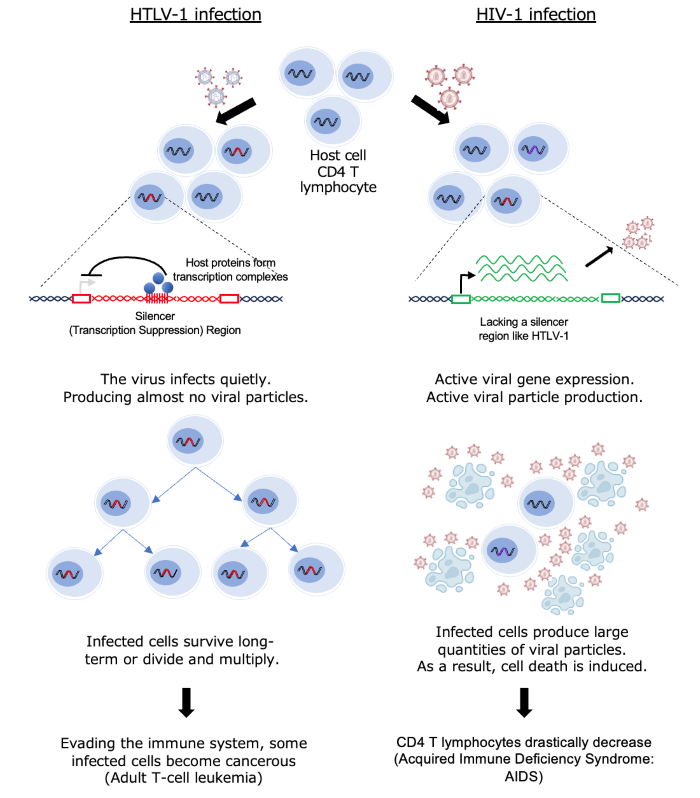
Human T-cell leukemia virus type 1 (HTLV-1) establishes long-term persistence by integrating into the host genome as a provirus. After a long incubation period, some infected individuals develop diseases such as adult T-cell leukemia (ATL). Although HTLV-1 encodes highly immunogenic gene products (e.g., Tax), viral expression is often constrained in vivo, helping infected cell clones evade immune surveillance. The molecular mechanisms that actively maintain this low-expression state (latency) have remained a central question.
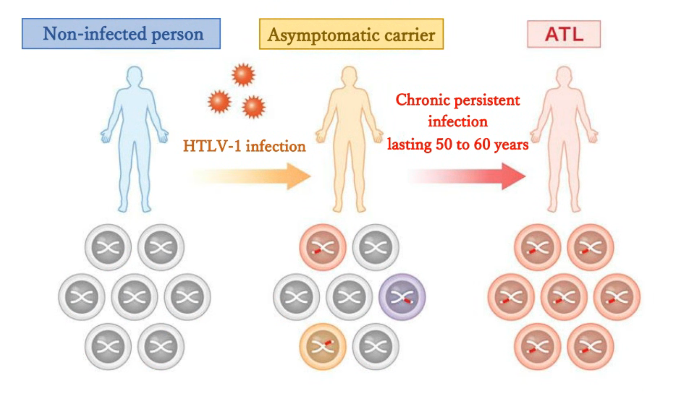
Summary
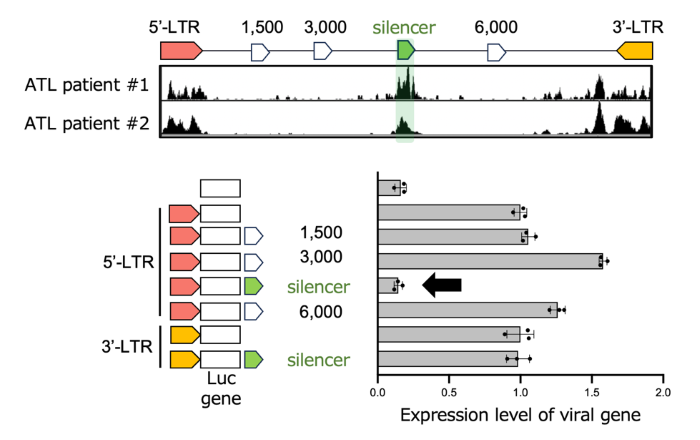
Using chromatin and functional analyses of patient-derived material and experimental systems, we identified an open chromatin region (OCR) within the HTLV-1 proviral genome (overlapping the pol region). Functional assays demonstrated that this region suppresses 5′LTR-driven sense transcription, revealing an intragenic “viral silencer” that helps enforce latency.
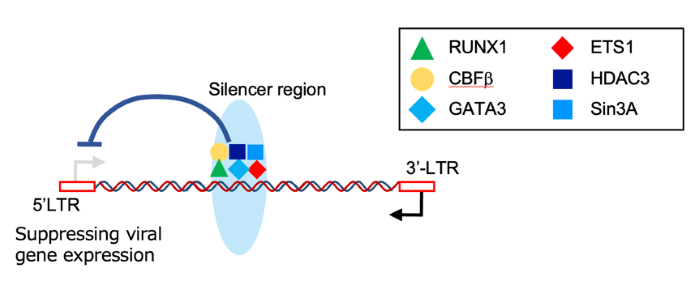
Mechanistically, we show that the host RUNX complex is recruited to this silencer and restrains viral transcriptional bursting, providing a direct link between host transcription factor occupancy and proviral quiescence.
Main findings
1. Identification of an intragenic viral silencer
A proviral internal OCR acts as a cis-acting element that suppresses HTLV-1 sense transcription from the 5′LTR.
2. RUNX complex recruitment and repression of transcriptional bursts
RUNX complex binding at the silencer reduces HTLV-1 transcriptional activity, supporting a model in which latency is actively maintained.
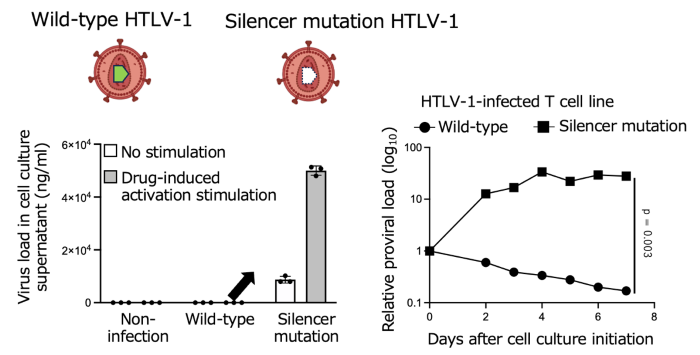
3. Phenotypic consequences of silencer disruption
Silencer mutations increase viral replication/production and are consistent with reduced latency maintenance, implying greater visibility to immune surveillance.
4. Transferability to HIV-1
Introducing the HTLV-1 silencer module into HIV-1 decreases virus production, indicating that cis-regulatory modules can reshape infection dynamics across retroviruses. These results provide a new perspective that may explain differences in infection dynamics among retroviruses.
Significance and outlook
These results suggest that HTLV-1 encodes a dedicated cis-regulatory silencer within its own genome to suppress transcription and support immune evasion during chronic infection. The RUNX–silencer axis provides a conceptual framework for manipulating proviral expression—either to promote controlled reactivation and immune clearance, or to reinforce suppression depending on therapeutic goals.
Article information
- Journal : Nature Microbiology
- Title : Intragenic viral silencer element regulates HTLV-1 latency via RUNX complex recruitment
- Authors : Kenji Sugata, Akhinur Rahman, Koki Niimura, Kazuaki Monde, Takaharu Ueno, Samiul Alam Rajib, Mitsuyoshi Takatori, Wajihah Sakhor, Md Belal Hossain, Sharmin Nahar Sithi, M. Ishrat Jahan, Kouki Matsuda, Mitsuharu Ueda, Yoshihisa Yamano, Terumasa Ikeda, Takamasa Ueno, Kiyoto Tsuchiya, Yuetsu Tanaka, Masahito Tokunaga, Kenji Maeda, Atae Utsunomiya, Kazu Okuma, Masahiro Ono & Yorifumi Satou
- Publication date : 13 May 2025
- DOI 1038/s41564-025-02006-7
