Circulating Cell-Free DNA of Bovine Leukemia Virus as a Biomarker for Enzootic Bovine Leukosis
Key Points
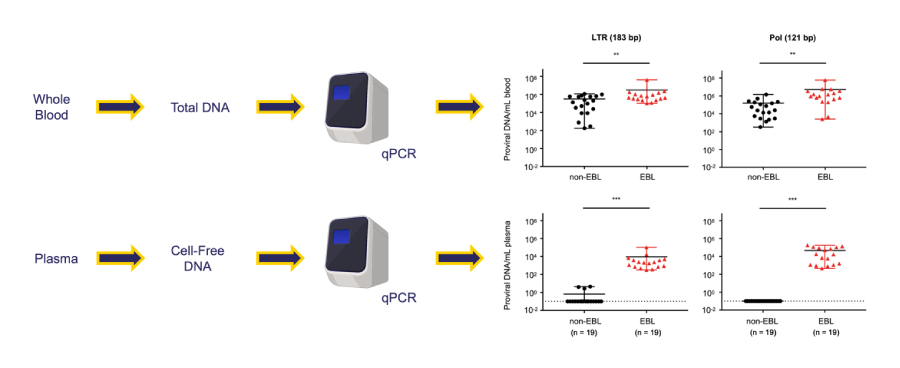
- Plasma-derived BLV circulating cell-free DNA (cfDNA) clearly distinguishes EBL from non-EBL cattle with 100% sensitivity and 100% specificity in receiver operating characteristic (ROC) analysis.
- In contrast to whole-blood PVL, plasma PVL demonstrates complete diagnostic separation between malignant and non-malignant cases.
- Detection sensitivity in plasma is critically dependent on PCR amplicon length, highlighting an essential technical parameter for cfDNA-based diagnostics.
- Comparative full-length viral genome sequencing revealed high concordance between lymphoma tissue and plasma cfDNA, indicating a shared molecular origin.
- Analysis of unique viral integration sites demonstrated that plasma BLV cfDNA predominantly originates from dominant tumor clones rather than peripheral blood mononuclear cells (PBMCs).
- Plasma BLV cfDNA therefore functions as a tumor-derived viral DNA biomarker that directly reflects clonal expansion and tumor burden.
Research Summary
EBL represents the malignant end-stage of BLV infection and continues to impose significant economic losses in the global cattle industry. Despite widespread BLV prevalence, progression to lymphoma occurs only in a minority of infected animals following years of clinical latency. The absence of a highly accurate diagnostic biomarker capable of distinguishing malignant transformation from asymptomatic infection has limited early intervention and optimal herd management strategies.
To overcome the diagnostic limitations of whole-blood proviral load measurement, this study investigated whether BLV-derived circulating cell-free DNA in plasma could serve as a tumor-specific biomarker. Quantitative PCR targeting the LTR and pol regions revealed that, whereas whole-blood PVL displayed overlapping distributions between groups, plasma PVL achieved complete diagnostic discrimination. ROC analysis confirmed perfect sensitivity and specificity, underscoring the robustness of this approach.
Mechanistically, molecular characterization demonstrated that plasma BLV cfDNA is not merely a reflection of infected peripheral blood cells. Instead, high sequence concordance between lymphoma tissue and plasma cfDNA, combined with shared viral integration site profiles, confirmed that circulating viral DNA predominantly originates from clonally expanded tumor cells. This establishes plasma BLV cfDNA as a bona fide tumor-derived biomarker rather than a surrogate of systemic infection.
Collectively, these findings redefine molecular diagnostics for EBL. By directly capturing tumor-derived viral DNA fragments released into circulation, plasma BLV cfDNA provides a minimally invasive, highly sensitive, and highly specific tool for early detection and disease stratification. Implementation of this biomarker in clinical and herd-level surveillance programs has the potential to enhance decision-making, reduce economic losses in the dairy and beef industries, and improve long-term BLV control strategies.
Article Information
Title: Circulating Cell-Free DNA of Bovine Leukemia Virus: A Promising Biomarker for Enzootic Bovine Leukosis
Authors: Jahan MI, Inenaga T, Makimoto S, Hossain MB, Matsuoka Y, Sithi SN, Rajib SA, Ansori ANM, Sugata K, Imakawa K, Kobayashi T, Satou Y
Journal: Microbiology and Immunology
DOI: 10.1111/1348-0421.13231
